Prof.Ye Sheng's Team Unlocks key to Tumor Growth
Malignant tumor cells are notorious for their ability to hijack the resources of human body, and to create toxic environments disrupting normal cellular function that fuel disease. Now, a research team led by Professor Sheng Ye at the School of Life Sciences has uncovered a crucial mechanism that cancer cells use to survive and thrive, offering new insights into potential anti-cancer therapies.
Cancer cells rely on a metabolic strategy called the Warburg effect, favoring glycolysis for energy production even when oxygen is available. This process generates excessive lactic acid, which must be expelled to prevent toxic buildup within the cell. The monocarboxylate transporter 1 (MCT1) plays a pivotal role in this process by shuttling lactic acid and protons out of cancer cells. However, this mechanism not only sustains tumor survival but also acidifies the surrounding microenvironment, suppressing immune responses and promoting cancer progression.
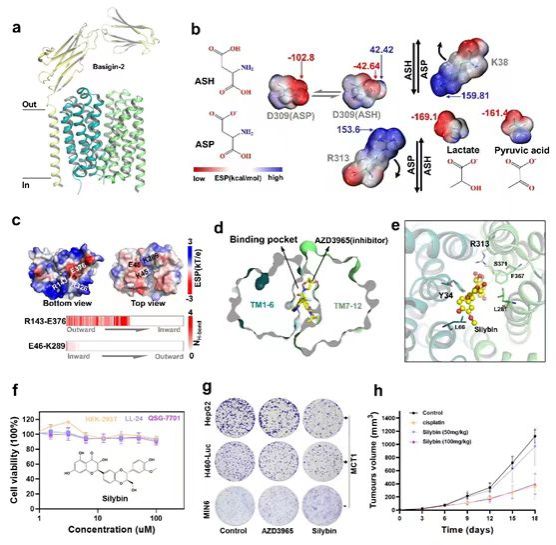
To better understand MCT1’s function, Professor Ye’s team employed advanced molecular simulations and experimental techniques. The results revealed that MCT1 relies on precise amino acid interactions to transition between inward- and outward-facing conformations, enabling efficient lactic acid transport . This dynamic process is driven by a coordinated "proton pump" mechanism, highlighting MCT1’s critical role in cancer cell metabolism.
Leveraging these insights, they further conducted virtual drug screening to identify potential inhibitors of MCT1. Among the tested molecules, silybin, a natural compound derived from traditional Chinese medicine, emerged as a potent and selective MCT1 blocker. By binding to MCT1’s core pocket, silybin effectively halts lactic acid export, leading to toxic accumulation within cancer cells. Laboratory experiments confirmed that silybin inhibits tumor growth in both cell cultures and animal models, highlighting its potential as a promising anti-cancer agent.
Published in Acta Pharmacologica Sinica, this study marks a major breakthrough in cancer metabolism and targeted drug development. By revealing the precise mechanism of MCT1’s function and identifying silybin as a selective natural inhibitor, this research opens a door towards innovative, safer and more effective cancer treatments.
“This research not only deepens our understanding of cancer metabolism but also lays the groundwork for developing next-generation cancer therapies” said Professor Sheng Ye. “We aim to deliver safer and effective treatment options to cancer patients in the near future.”
By Eva Yin

